Solved 1. A cup of hot 50°C water is poured onto 35g of ice

By A Mystery Man Writer
Answer to Solved 1. A cup of hot 50°C water is poured onto 35g of ice

Solved 1. A cup of hot 50°C water is poured onto 35g of ice
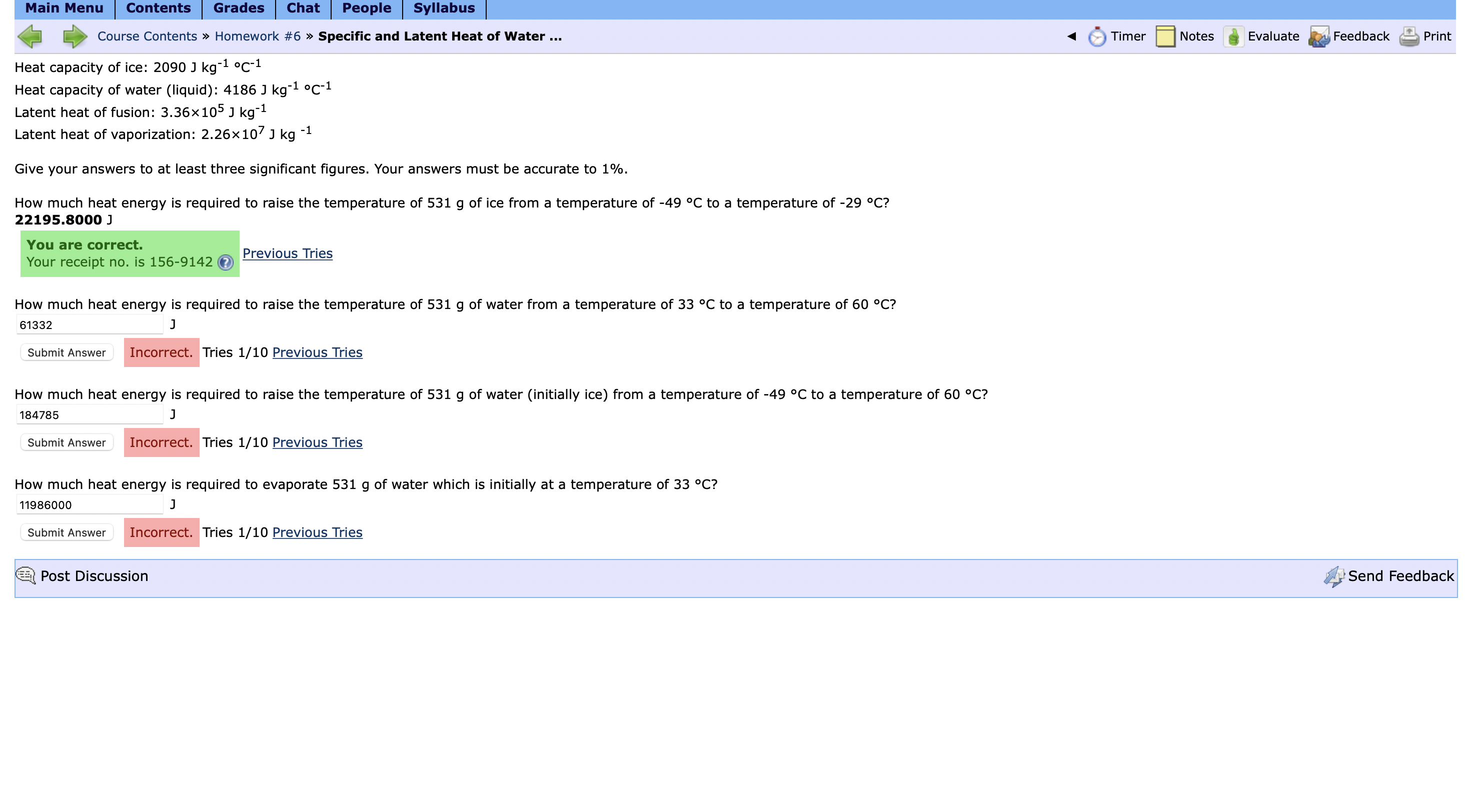
Solved Heat capacity of ice: 2090Jkg-1°C-1Heat capacity of
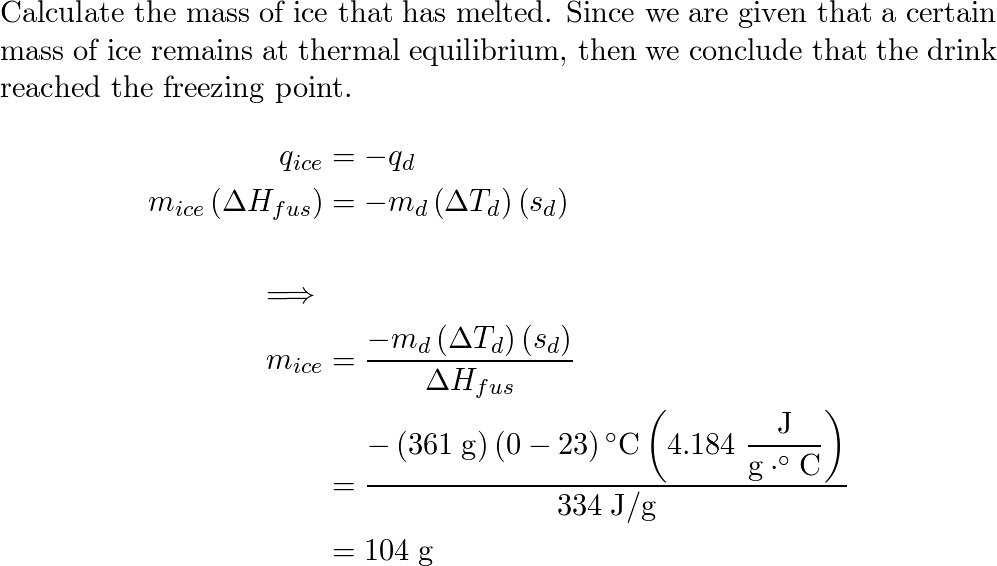
Ice at 0 °C is placed in a Styrofoam cup containing 361 g of

Solved 1. A cup of hot 50°C water is poured onto 35g of ice

Calorimetry - Chemistry

Ontario occurrences of float, placer gold and other - Geology Ontario

First for Women January 16, 2023 (Digital)

A calorimeter of mass 50 g and specific heat capacity 0.42 J {g}^{-1} {℃ }^{ -1 } contains some mass of water {20}^{o}C. A metal piece of mass 20 g {100}^{o}C is
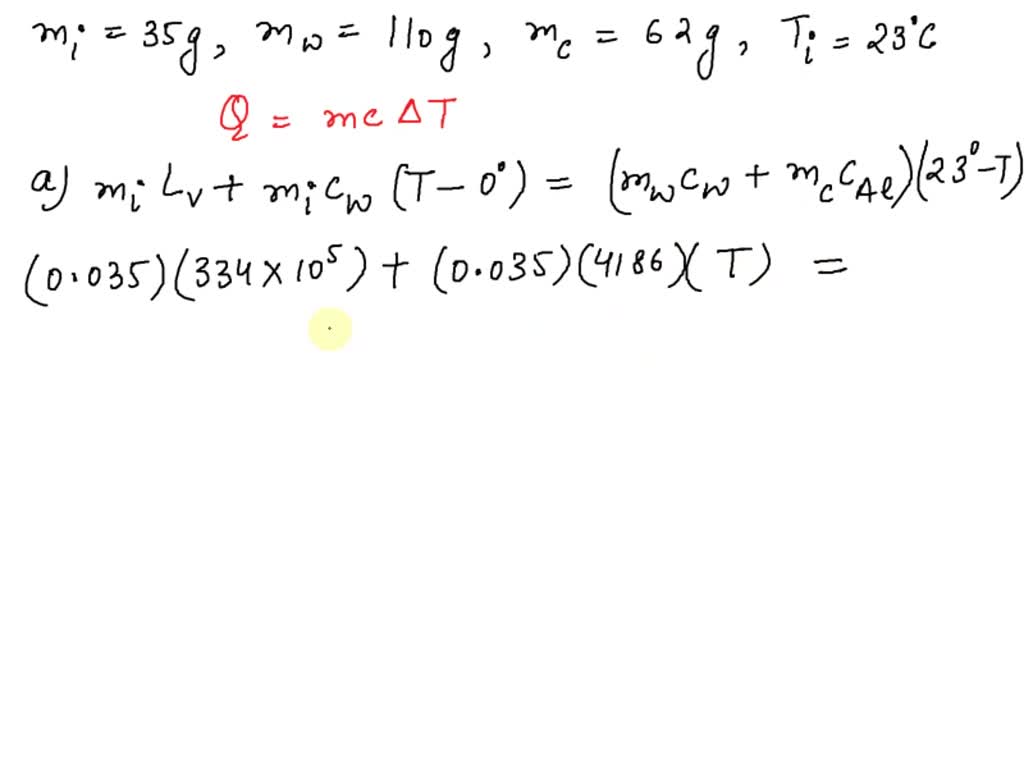
SOLVED: A 35-g ice cube at 0.0 °C is added to 110 g of water in a 62-g aluminum cup. The cup and the water have an initial temperature of 23 °C. (
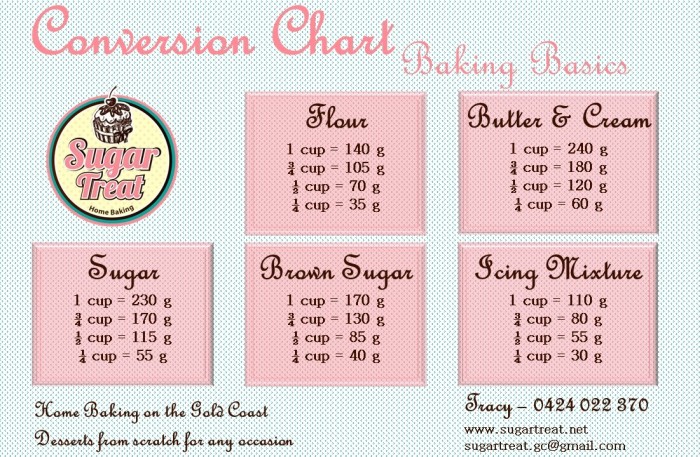
- Ingredients – Sugar Treat – Home Baking on the Gold Coast
- Examples of protein sources for athletes (or practical meals for older

- Labrada Lean Body Hi-Energy Meal Replacement Shake, Power Latte, 35g Protein, 2.47lb

- Egg white scramble. 241 calorie breakfast 35 grams of protein ❤️ : r/1200isplenty
- SOLVED: A 25 gram piece of hot metal at 97°C is plunged into a 35
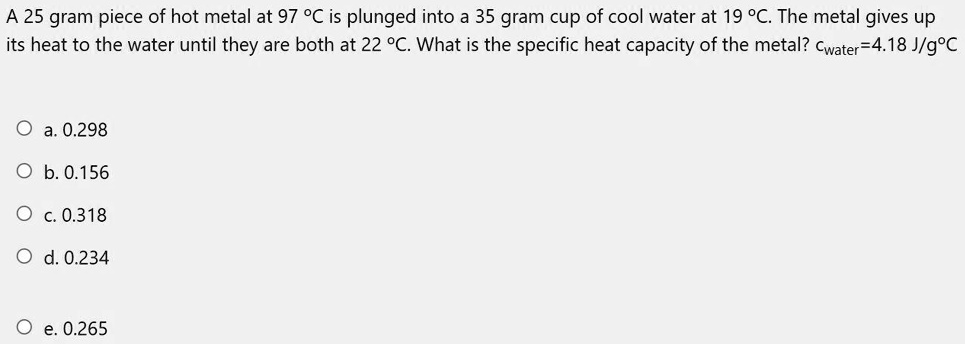
- Something Blue Lace Bridal Underwear, Just Married Hipster, Cheeky Lace Bride Hot Short, Bridal Lingerie for the Honeymoon, Mrs Lace Hipster - Canada

- What is the difference between a country road and regular road? - Quora
- Pace International Kabaddi Shoes Boxing & Wrestling Shoes For Men

- Orangetheory Gym Near You In Manhattan - Midtown West, NY

- Calzedonia Meia Calça Total Shaper Fio 50 - Marrom
