The vapour pressure of a solution having 2.0 g of solute X (gram
By A Mystery Man Writer
The vapour pressure of a solution having 2.0 g of solute X (gram atomic mass=32 g/mol) in 100 g of CS2 (vapour pressure =854torr) is 848.9 torr.The molecular formula of solute 1) X 2)X2 3)X4 4)X8
The vapour pressure of a solution having 2-0 g of solute X -gram atomic mass-32 g-mol- in 100 g of CS2 -vapour pressure -854torr- is 848-9 torr-The molecular formula of solute 1- X 2-X2 3-X4 4-X8

Chapter 16 Colligative properties of solutions

14. The vapour pressure of the solution having 2.0 g of solute (a molecu the solution having 2.0 g of solute (a molecule of x with atomic ma = 32 g/mol) in

At 48°C the vapour pressure of pure CS, is 850 torr. A solution of 2.0 g of sulphur in 100 g of CS2 has a vapour pressure 844.9 torr. Determine the atomicity

PPT - Chapter 7: Solutions and Colloids PowerPoint Presentation

Answered: 18) Predict the vapor pressure of a…

Solutions Homework Help, Questions with Solutions - Kunduz
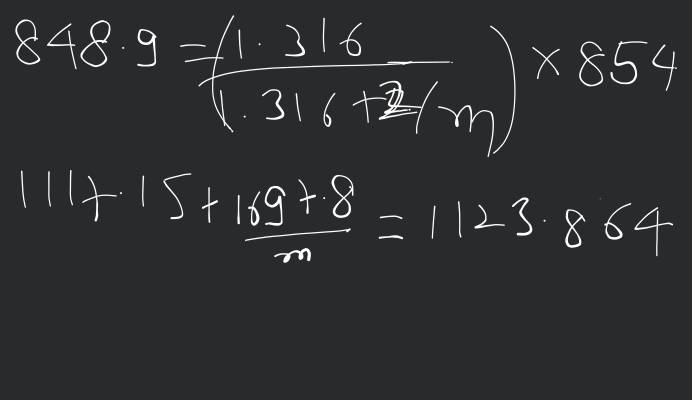
The vapour pressure of a solution having 2.0 g of a solute X( molar mass ..

How to solve this question, I used the formula P -Ps/Ps, as masses are given I am not - Chemistry - - 14746401

SOLVED: The vapour pressure of a solution having 2.0 g of solute X
- Quest Tortilla Style Protein Chips Nacho Cheese, 32 g

- Pulseira Trançada Ouro 18k 750 32 G – Ourique Joalheria

- TRENTO CHOCOLATE WAFER DARK 32 G x 1
- Smartphone Motorola Moto G4 Plus Dual Chip Tela de 5.5 4G 32 GB

- Fralda BabySec Ultra Mega 6 Pacotes Tamanho G - 32 Unidades Cada - Fralda Descartável - Magazine Luiza




